# How to use custom primers on the NovaSeq X Series
**Background**
The NovaSeq X Series supports Custom Primers using three available Custom Primer (CP) wells on the reagent cartridge. The custom primers will be diluted in the NovaSeq X Series Custom-Primer buffer, available from the [NovaSeq X Series Reagents Kits](https://www.illumina.com/products/by-type/sequencing-kits/cluster-gen-sequencing-reagents/novaseq-x-series-reagent-kits.html) page on the Illumina website. Sequencing run setup and limitations will vary depending on Control Software version, see below for details.
**Important Notes**
*For NovaSeq X Series Control Software v1.3 and above:*
Select the applicable custom primer checkboxes during run setup to inform the custom primer well position during the sequencing run. The custom primer recipe is no longer required to use custom primers on the NovaSeq X Series, and uploading a custom recipe will disable the custom primer checkboxes.
*For NovaSeq X Series Control Software v1.2.2 and below:*
Download the appropriate custom primer recipe XML file from the [NovaSeq X Plus Product Files](https://support.illumina.com/sequencing/sequencing_instruments/novaseq-x-novaseq-x-plus/product-files.html) page and upload the custom primer recipe during run review. If a custom recipe is not available in the desired read configuration, Illumina recommends spiking the custom primer into the Illumina primer wells. See Knowledge Article [Spiking custom primers into the Illumina sequencing primers](https://knowledge.illumina.com/library-preparation/general/library-preparation-general-reference_material-list/000001542) and section '**How to spike in custom primers into Illumina Primer Wells on the NovaSeq X Series**' for additional information.
**Note on PhiX**:
When custom primers are used for Read 1 or Read 2, the software directs the instrument to pull from the CP1 and CP2 wells. Therefore, Illumina primers pre-loaded into the reagent are not used for the sequencing run. If Illumina primers are not used for Read 1 or Read 2, the optional Illumina PhiX control will not be sequenced. To use the PhiX control with custom primers, see section '**How to spike in custom primers into Illumina Primer Wells on the NovaSeq X Series**' below or contact [Illumina Technical Support](https://www.illumina.com/company/contact-us.html) for guidance.
**Note on Lanes**:
When custom primers are used on the NovaSeq X Series, the custom primer is applied to the entire flow cell and cannot be selected for individual lanes.
**Prepare the Custom Primers:**
1. If the custom primers are stored frozen, thaw them at room temperature and then store on ice.
2. Dilute the primers to a final concentration of **0.3 µM** using the **NovaSeq X Series Custom-Primer buffer**. For the primer volumes, see **Table 1**.
3. When combining primers to make the custom Index Read or any custom read primer mixture, the final concentration of **each** primer must be 0.3 µM. This will result in a higher total primer concentration and is expected.
1. **Example**: For Index 1 and Index 2 primer mix, prepare each primer solution at 0.6 µM, then combine 3.5 ml of each primer solution for a final volume of 7.0 ml with each primer at 0.3 µM and a total concentration of 0.6 µM.
**Table 1**: Custom Primer well positions and volumes in milliliters (ml)
| Custom Primer | Well Position | Volume (ml) for 10B and 25B Flow Cell | Volume (ml) for 1.5B Flow Cell | Conc. for each custom primer (µM) |
| ------------------------------- | ------------- | ------------------------------------- | ------------------------------ | --------------------------------- |
| Custom Read 1 Primer | CP1 | 5.0 | 3.0 | 0.3 |
| Custom Read 2 Primer | CP2 | 5.0 | 3.0 | 0.3 |
| Custom Index 1 and 2 Primer Mix | CP3 | 6.0 | 5.0 | 0.3 |
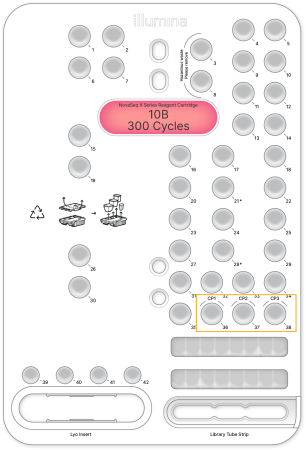
**Add Custom Primers to the Reagent Cartridge:**
1. Use a low-lint tissue to wipe clean the foil seal covering each custom primer position listed in **Table 1**.
2. Using a clean pipette tip, pierce the foil seal covering each custom primer position.
3. Add the appropriate volumes of custom primer to the correct position on the reagent cartridge.
* Avoid touching the foil seal when dispensing the primer.
**Setup sequencing run to use Custom Primers**\*\*:\*\*
Using custom primers is specified during run setup either by selecting checkboxes or by a custom recipe.
*For NovaSeq X Series Control Software v1.3 and above:*
1. After selecting the appropriate run, select the appropriate Custom Primers checkboxes on the '**Review your run information**' page.
2. After reviewing the run setup information, select **Load consumables** to proceed.

*For NovaSeq X Series Control Software v1.2.2 and earlier:*
After selecting the appropriate run, add a custom recipe file to the run on the '**Review your run information**' page during run setup.
1. Navigate to the [NovaSeq X Plus Product Files](https://support.illumina.com/sequencing/sequencing_instruments/novaseq-x-novaseq-x-plus/product-files.html) Support Page and download the Custom Primer Recipe XML file.
2. Save the Custom Primer Recipe XML file to the instrument, typically to the **Desktop** or **Downloads** directory.
3. In the **Review your run information** screen, select **Select file...** under Custom Recipe (optional).
4. Navigate to the directory where the XML file was saved on the instrument, then select the custom recipe XML file.
5. After reviewing the run setup information, select **Load consumables** to proceed.
**How to spike in custom primers into Illumina Primer Wells on the NovaSeq X Series:**
The NovaSeq X/X Plus uses two primer solutions that combine primers for Read 1 and 2, and Index 1 and 2. If custom primers are only required for specified Reads, spike-in the concentrated primers into the appropriate wells for the Reads that require custom primers. When spiking in custom primers, **no changes** to the run set up are needed and a custom recipe is not required.
**Table 2**. provides the well positions, Illumina primer volume, and the recommended custom primer (CP) volume to add in microliters (µl) **assuming a starting concentration of 100 micromolar (µM) for the custom primer stock that will be used.**
**Table 2**.
| Kit Type | Primer Mix | Reads Primed | Cartridge Position | Primer Fill Vol. (ml) | Final Conc. (µM) | CP Conc. (µM) | CP Vol. (µl) |
| -------- | ---------- | ------------ | ------------------ | --------------------- | ---------------- | ------------- | ------------ |
| **1.5B** | TP21 | Read 1/2 | 28 | 4.6 | 0.3 | 100 | 13.8 |
| | TP14 | Index 1/2 | 21 | 4.7 | 0.3 | 100 | 14.1 |
| | | | | | | | |
| **10B** | TP21 | Read 1/2 | 28 | 7.7 | 0.3 | 100 | 23.1 |
| | TP14 | Index 1/2 | 21 | 6.9 | 0.3 | 100 | 20.7 |
| | | | | | | | |
| **25B** | TP21 | Read 1/2 | 28 | 8.1 | 0.3 | 100 | 24.3 |
| | TP14 | Index 1/2 | 21 | 6.9 | 0.3 | 100 | 20.7 |
\
\
\
| |
| :--------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------: |
| *For any feedback or questions regarding this article (Illumina Knowledge Article #7779), contact Illumina Technical Support* [*techsupport@illumina.com*](mailto:techsupport@illumina.com?subject=Question%2FFeedback%20Regarding%20Illumina%20Knowledge%20Article%20#000007779%20-%20Instrumentation%20\&body=Dear%20Illumina%20Technical%20Support,%0D%0A%0D%0A)*.* |
---
# Agent Instructions: Querying This Documentation
If you need additional information that is not directly available in this page, you can query the documentation dynamically by asking a question.
Perform an HTTP GET request on the current page URL with the `ask` query parameter:
```
GET https://knowledge.illumina.com/instrumentation/novaseq-x-x-plus/instrumentation-novaseq-x-x-plus-reference_material-list/000007779.md?ask=
```
The question should be specific, self-contained, and written in natural language.
The response will contain a direct answer to the question and relevant excerpts and sources from the documentation.
Use this mechanism when the answer is not explicitly present in the current page, you need clarification or additional context, or you want to retrieve related documentation sections.